
Incidence of grade 3-5 adverse events was similar in the pembrolizumab-combination (71.9%) and placebo-combination (66.8%) groups.įirst-line pembrolizumab plus pemetrexed-platinum continued to demonstrate substantially improved OS and PFS in metastatic nonsquamous NSCLC, regardless of PD-L1 expression or liver/brain metastases, with manageable safety and tolerability.KENILWORTH, N.J.-( BUSINESS WIRE)-Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced positive results from two studies from the company’s leading lung cancer research program. 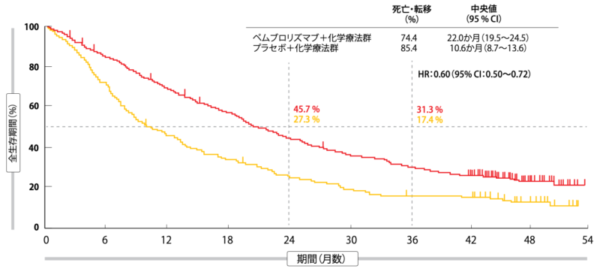
OS and PFS benefits with pembrolizumab were observed regardless of PD-L1 expression or presence of liver/brain metastases. Median (95% CI) time from randomization to objective tumor progression on next-line treatment or death from any cause, whichever occurred first (progression-free-survival-2 PFS-2) was 17.0 (15.1 to 19.4) months and 9.0 (7.6 to 10.4) months, respectively (HR, 0.49 95% CI, 0.40 to 0.59). No alpha was assigned to this updated analysis.Īs of Septem(median follow-up, 23.1 months), the updated median (95% CI) OS was 22.0 (19.5 to 25.2) months in the pembrolizumab-combination group versus 10.7 (8.7 to 13.6) months in the placebo-combination group (hazard ratio, 0.56 95% CI, 0.45 to 0.70]). Response was assessed per RECIST (version 1.1) by central review. Eligible patients with disease progression in the placebo-combination group could cross over to pembrolizumab monotherapy. Patients were randomly assigned (2:1) to receive pemetrexed and platinum plus pembrolizumab (n = 410) or placebo (n = 206) every 3 weeks for 4 cycles, then pemetrexed maintenance plus pembrolizumab or placebo for up to a total of 35 cycles. We report an updated analysis from KEYNOTE-189 (: NCT02578680). In KEYNOTE-189, first-line pembrolizumab plus pemetrexed-platinum significantly improved overall survival (OS) and progression-free survival (PFS) compared with placebo plus pemetrexed-platinum in patients with metastatic nonsquamous non‒small-cell lung cancer (NSCLC), irrespective of tumor programmed death-ligand 1 (PD-L1) expression. 24 Fondazione Istituto di Ricovero e Cura a Carattere Scientifico Istituto Nazionale dei Tumori, Milan, Italy.21 Kansai Medical University Hospital, Osaka, Japan.20 Chris O'Brien Lifehouse, Camperdown, New South Wales, Australia.19 Hospital Universitario Quirónsalud Madrid, Madrid, Spain.18 Department of Oncology, University of Turin, Azienda Ospedaliero-Universitaria San Luigi, Orbassano, Italy.17 David Geffen School of Medicine at University of California Los Angeles, Los Angeles, CA.16 LungenClinic, Airway Research Center North, German Center for Lung Research, Grosshansdorf, Germany.15 Epworth Healthcare, Richmond, Victoria, Australia.14 Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, Milan, Italy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
